Rajca Research Group
Organic, Polymer and Biomaterials Chemistry
Rational Designs | Syntheses | Measurements


Design and synthesis of high-spin organic molecules and polymers are among important aspects in the development of novel magnetic materials, particularly organic magnets. The molecular designs are aimed at π-conjugated organic molecules with half-filled topologically degenerated or nearly degenerated non-bonding molecular orbitals (NBMOs). Such degeneracies are usually required (but not always sufficient) for a molecule to possess high-spin ground state.
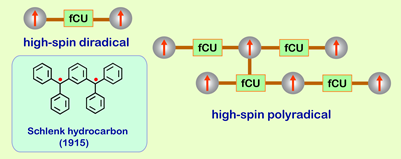 The design strategy for high-spin polyradical is based upon connecting spin sites for an unpaired electron (free radical) and ferromagnetic coupling units (fCUs). Two spin sites connected to an fCU give rise to a high-spin diradical with S = 1 ground state. An example is the Schlenk hydrocarbon, in which a 1,3-phenylene fCU links two arylmethyl spin sites. The conceptual extension to high-spin polyradical relies on connecting, in an alternating mode, the fCUs and spin sites.
The design strategy for high-spin polyradical is based upon connecting spin sites for an unpaired electron (free radical) and ferromagnetic coupling units (fCUs). Two spin sites connected to an fCU give rise to a high-spin diradical with S = 1 ground state. An example is the Schlenk hydrocarbon, in which a 1,3-phenylene fCU links two arylmethyl spin sites. The conceptual extension to high-spin polyradical relies on connecting, in an alternating mode, the fCUs and spin sites.
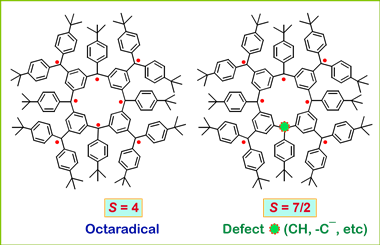
Our research group has prepared numerous high-spin organic polyradicals, including the dendritic and the star-branched structures. In these polyradicals, there is only one pathway for spin coupling. If there is a defect, i.e. a failure to generate an unpaired electron, at one of the non-terminal spin sites, the ferromagnetic spin coupling will be interrupted, leading to a mixture of spin systems with low values of S. Therefore, average values of S for these radicals were limited to S = 5 or less.
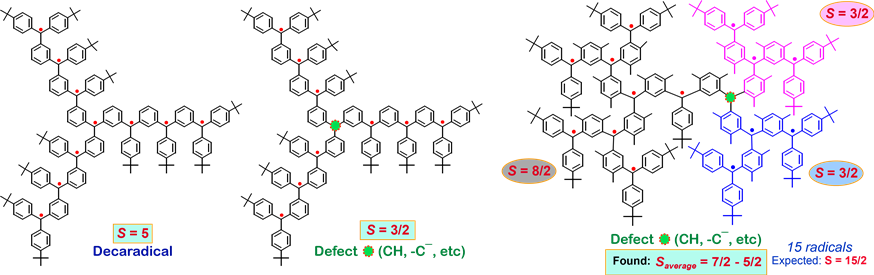
To solve this problem, the macrocyclic polyradicals were prepared. The cyclic structure provides two pathways for spin coupling. One defect would still result in one spin system with only 1/2 unit less in the value of S.
 To obtain very large values of S, a polyradical must contain large number of the spin sites. As the number of the spin sites increases, the probability of having two or more defects becomes greater Molecular designs of the dendritic-macrocyclic and macrocyclic-macrocyclic structures are to maximize the number of spin sites and to avoid problems with the defects. In order to construct these novel molecules, the spin cluster approach was developed. With the dendritic-macrocyclic design strategy, the highest spin organic molecule to date with S = 10 was prepared by our research group. This molecule has an overall shape of a prolate ellipsoid with diameter of 4 and 2 nm, as determined by Monte Carlo conformational search and SANS (Small Angle Neutron Scattering) experiments.
To obtain very large values of S, a polyradical must contain large number of the spin sites. As the number of the spin sites increases, the probability of having two or more defects becomes greater Molecular designs of the dendritic-macrocyclic and macrocyclic-macrocyclic structures are to maximize the number of spin sites and to avoid problems with the defects. In order to construct these novel molecules, the spin cluster approach was developed. With the dendritic-macrocyclic design strategy, the highest spin organic molecule to date with S = 10 was prepared by our research group. This molecule has an overall shape of a prolate ellipsoid with diameter of 4 and 2 nm, as determined by Monte Carlo conformational search and SANS (Small Angle Neutron Scattering) experiments.
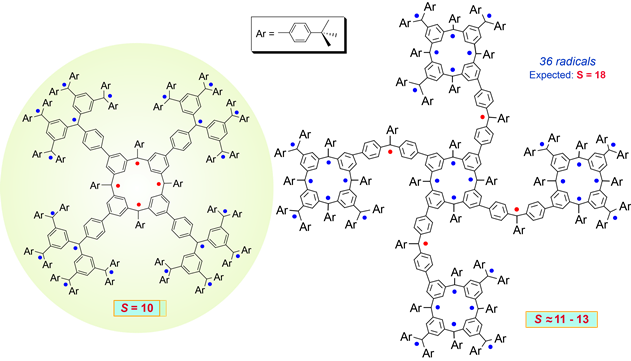 The macrocyclic-macrocyclic molecular design provide a polyradical with 36 sites for unpaired electrons. Very high spin polyradicals with well-defined fractal dimension may serve as models for understanding of magnetic phenomena on mesoscopic scale. Such molecules, with Mw of about 10,000 Da, possess interesting 3-D shapes, like small proteins of similar size.
The macrocyclic-macrocyclic molecular design provide a polyradical with 36 sites for unpaired electrons. Very high spin polyradicals with well-defined fractal dimension may serve as models for understanding of magnetic phenomena on mesoscopic scale. Such molecules, with Mw of about 10,000 Da, possess interesting 3-D shapes, like small proteins of similar size.
Elaboration of the spin cluster approach leads to a polymacrocyclic highly cross-linked π-conjugated polymer network polyradical with multiple spin coupling pathways. The network consists of S = 2 macrocyclic calix[4]arene modules, which are cross-linked with S =1/2 connecting modules. High density of macrocycles in the network should address the problem with defects. Also, the alternating conectivity of two types of radical modules with unequal spins (ferromagnetic/ferrimagnetic arrangement of spin) should facilitate large net S values for either ferromagnetic or antiferromagnetic coupling between the modules. This feature is important for polymers where steric hindrance may lead to out-of-plane twisting within the π-conjugated system.
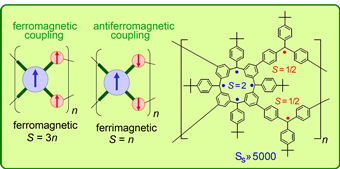 The minor organic soluble product of the network (Mw = 3 - 5 x 105 Da) gave paramagnetic polymers with an average S > 40 (J. Am. Chem. Soc, 1999, 121, 6308.) The major organic insoluble, highly cross-linked, products gave polymer with very large average S of about 5000; slow reorientation of the magnetization by a small magnetic field (1 Oe) below 10K were observed. Magnetic behavior of the polymer is comparable to that of insulating spin glass and blocked superparamagnets (Science, 2001, 294, 1503). This is the first report of a π-conjugated organic polymer with such magnetic properties. This result shows that organic polymer magnets (proposed by Mataga in 1968) can be prepared.
The minor organic soluble product of the network (Mw = 3 - 5 x 105 Da) gave paramagnetic polymers with an average S > 40 (J. Am. Chem. Soc, 1999, 121, 6308.) The major organic insoluble, highly cross-linked, products gave polymer with very large average S of about 5000; slow reorientation of the magnetization by a small magnetic field (1 Oe) below 10K were observed. Magnetic behavior of the polymer is comparable to that of insulating spin glass and blocked superparamagnets (Science, 2001, 294, 1503). This is the first report of a π-conjugated organic polymer with such magnetic properties. This result shows that organic polymer magnets (proposed by Mataga in 1968) can be prepared.
Following the design principles developed for the polyarylmethyls, we are working on syntheses of high spin polynitroxides and other polyradicals stable at ambient condition. We are in search for promising building blocks for preparation of organic polymer magnets with improved ordering temperature and stability at ambient conditions ("plastic magnets"). Follow this link
SANS Experiments at the NIST Center for Neutron Research, Gaithersburg, Maryland
We thank the NIST Center for Neutron Research (NCNR) for the neutron beam time and
Dr. Dan Dender (Research Facilities Operations Group, NCNR) for his help with the cryostat and the photos.
Collaborators: Dr. Paul Butler and Dr. Sungmin Choi (Neutron Condensed Matter Science Group, NCNR)
S. Rajca, A. Rajca, J. Wongsriratanakul, P. Butler, S. Choi, Organic Spin Clusters. Dendritic-Macrocyclic Polyarylmethyl Polyradical with Very High-Spin of S = 10 and its Derivatives: Synthesis, Magnetic Studies, and Small Angle Neutron Scattering, J. Am. Chem. Soc., 2004, 126, 6972 – 6986.